File:TACG A 178546 O F0001g.jpg
Jump to navigation
Jump to search
Size of this preview: 605 × 599 pixels. Other resolutions: 242 × 240 pixels | 485 × 480 pixels | 750 × 743 pixels.
Original file (750 × 743 pixels, file size: 259 KB, MIME type: image/jpeg)
Summary
| Description |
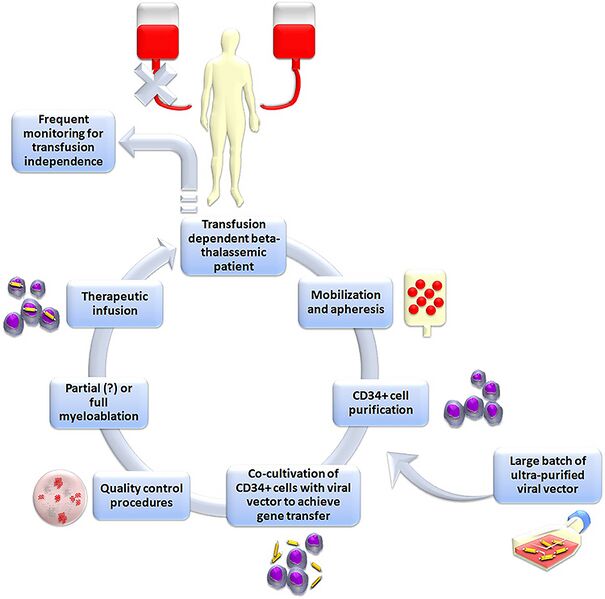
English: Figure 1 Schematic representation of the classic gene addition protocol for the gene therapy of beta-thalassemia. In brief, Plerixafor+granulocyte-colony stimulating factor (G-CSF) is administered to the patient in order to mobilize hematopoietic stem cells into peripheral blood. Peripheral blood mononuclear cells are then collected with leukapheresis and enriched in CD34+ hematopoietic stem cells. These cells are co-cultured with a viral vector designed to express normal human beta-globin, followed by quality control. Ultimately, the patient is subjected to myelosuppression and then engrafted with the gene-corrected cells. |
| Date | |
| Source | https://www.dovepress.com/gene-therapy-for-beta-thalassemia-updated-perspectives-peer-reviewed-fulltext-article-TACG |
| Author | Karponi G, Zogas N |
Licensing
English: This file is licensed CC BY-NC 3.0
This file was uploaded with UploadWizard.
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | Thumbnail | Dimensions | User | Comment | |
|---|---|---|---|---|---|
| current | 21:41, 26 December 2023 |  | 750 × 743 (259 KB) | Ozzie10aaaa (talk | contribs) | Uploaded a work by Karponi G, Zogas N from https://www.dovepress.com/gene-therapy-for-beta-thalassemia-updated-perspectives-peer-reviewed-fulltext-article-TACG with UploadWizard |
You cannot overwrite this file.
File usage
There are no pages that use this file.